doi: 10.15389/agrobiology.2012.1.72eng
УДК 635.21/.24:57.043:581.132
PHOTOSYNTHESIS, ASSIMILATES TRANSPORT AND PRODUCTIVITY IN POTATO PLANTS OF THE NEVSKII VARIETY GROWN UNDER DIFFERENT INSOLATION
V.I. Chikov1, G.A. Salyakhova1, G.F. Safiullina2, F.F. Zamalieva2
The authors investigated the effect of 25, 50 и 95 % insolation on 14СО2 assimilation and the distribution of photosynthetic 14C-products in potato plant’s organs. It was shown, that carboxylating activity in leaves (14СО2 absorption) was increased only in the conditions of 95 % insolation, but under 25 и 50 % insolation the significant differences were not revealed. The intensity of labeled assimilate export from the 14С-donor lamina was maximal during medium lighting and minimal — during saturated one. The 14С distribution in plant organs in the conditions of high-level lighting was characterized by rise of assimilates transport to crown of stem and by reduced entry of 14С to tubers. The efficiency of photosynthetic apparatus (the mass of tubers per mass unit of potato tops) in 50 and 95 % variants was the greatest and the least, respectively.
Keywords: photosynthesis, assimilate transport, Solanum tuberosum, photosynthetic productivity, radiation intensity.
Regulation of production processes in plants includes such important factor as donor-acceptor ratio (DAR) between photosynthetic organs and those utilizing assimilates. It is believed (1) that formation of a harvest (accumulation of photosynthesis products in economically important organs-acceptors) depends on the intensity of photosynthesis in leaves-donors, which, in turn, is activated by demands of accepting organs and tissues. DAR participants have different functions (2). Leaves are usually donors of photosynthesis products exported to utilizing organs (although young leaves utilize some part of assimilates).
Most of the published data on DAR describe experiments on plants with artificially disturbed relationship between the weight of donor and acceptor organs while maintaining, as a rule, the optimal conditions of photosynthesis in leaves. For example, reduced leaf area relative to the weight of accepting organs, or controversially.
At the same time, in nature, a photosynthetic apparatus usually operates under varying illumination. Studying the role of this factor in photosynthesis often was performed under short-term shifts in illumination discovering only fast regulatory mechanisms (changes in stomatal opening, supply of Calvin cycle pools, photorespiration activity, etc.).
So, it’s yet unknown how the production processes are regulated in plants exposed to constantly limited illumination. This problem is particularly important in respect to potato accumulating photosynthesis products mainly in underground organs - tubers and a suction zone of roots, which are two competing acceptors.
This experiment was performed in order to find out how the improved in vitro propagated potato plants use assimilates during an exposure to different illumination.
Technique. The study was performed on potato plants the variety Nevsky grown according to the original method developed in Tatarstan Research and Development Institute of Agriculture. In vitro propagated plants were grown in a gauzed frame to obtain microtubers. A greenhouse frame covered with cotton gauze provided sufficient ventilation and prevented the transfer of viruses by winged aphids. A surface of soil was strewed with a river sand to improve drainage and limit migration of other phytopathogens. In vitro propagated potato plants were planted in a protected ground in early June using polyethylene boxes of 70 × 30 cm with a bottom side of nonwoven acryl fabric. Soil composition: peat, river sand (2:1) and vermicompost (2 kg/m2). Each box contained about 1,5 kg soil per one plant. The plants were grown in three variants of different illumination - 25, 50 and 95% of total insolation. Most of the area inside the gauze frame was shaded by one layer of a cotton gauze (variant 50%), one section was covered with additional second layer of a cotton gauze in 30-40 cm above tops and from their sides (variant 25%; watering was carried out over the second layer of gauze) and another section was covered with a mosquito netting made of a synthetic material (variant 95%). The intensity of photosynthetically active solar radiation (PAR) on the day of the experiment was measured by the actinometer M-3 (Russia) with light filters. Without shading, it amounted to 400 W/m2, in the variants of 95, 50 and 25% insolation – respectively, 380, 200 and 100 W/m2.
The intensity of 14CO2 assimilation was determined during a mass flowering of plants on a sunny cloudless day (July 24) in a period from 1000 to 1200. A photosynthetic clip chamber was placed on the terminal leaflet of a middle tier leaf in each variant (25, 50 and 95% lightning) and 14СО2 was pumped through it from a gas-holder for 2,5 min as described (3). Carbon dioxide content in the gas-holder was about 0,03.%. 3 min later the leaflets were cut off and fixed with boiling 80% ethanol to determine the intensity of absorption of 14СО2 . After the introduction of 14СО2 , the plants were grown for 1 day in usual conditions while the labeled photosynthesis products were distributing in organs. The leaf through which 14СО2 had been introduced was marked for later identification. On the next day, the plants were divided into parts (a leaflet – donor of 14C-assimilates, the rest of a donor leaf, shoot apex with juvenile leaves, stems with leaves above and below a donor leaf, roots and tubers). All parts were fixed with boiling 80% ethanol. Fixed samples were homogenized with 60% ethanol and their total radioactivity was measured on a scintillation counter Delta-300 (“Tracor Analytic”, USA). Experiments with 14C were carried out in 5 biological replicates and 2 analytical repetitions.
Productivity characteristics were determined on September 14: weigh of leaves and tubers, number of tubers (in each variant - 10-12 plants from six boxes).
Mean values calculated per one plant (or leaf) with a standard error are shown in diagrams.
Results. Under a natural content of CO2, photosynthesis in potato plants attains saturation in the illumination of about half a natural solar radiation (4), so the experimental illumination corresponded to light deficit (25%), optimal conditions close to saturation (50%) and abnormal excess of light (95%).
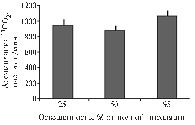 |
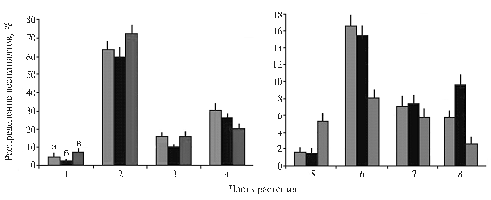
Fig. 1. Assimilation of 14СО2 in leaves of potato cv Nevsky grown in different light conditions |
The intensity of 14СО2 assimilation during 2,5 minutes wasn’t much different in plants grown in unequal light conditions (Fig. 1). A significant excess (12-18%) of this index was observed only in high illumination. Since the day of the experiment was hot and sunny, differences between the variants can be explained by the degree of stomatal opening. In the gauze frame, air flow (wind) was light, and even lighter in the variant of a minimum illumination shaded by the second layer of gauze. These conditions contributed to significant air humidity and a maximum opening of stomata. In contrast, plants grown in high light were strongly heated and a part of the stomata were probably closed.
|
Fig. 2. Distribution of 14С-products in 24 h after assimilating 14СО2 by the terminal leaflet of a medium tier leaf in plants of potato cv Nevsky grown in different light conditions: a, б, в — respectively, 25, 50 and 95 % of total insolation; 1 — stem segment above 14С-donor leaf, 2 — terminal leaflet of 14С-donor leaf exposed to 14СО2 3 — the rest of 14С-donor leaf, 4 — aboveground segment of a stem below 14С-donor leaf, 5 — shoot apexes, 6 — tubers, 7 — roots, 8 — ratio of radioactivity in plant parts above and below the donor leaf Denotations: |
After a short-term exposure of a leaf to 14СО2, resulting radioactivity reflects only the amount of absorbed label. Final photosynthesis products keep a lower content of the labeled carbon since a significant share of 14С (30-50%) is metabolized in photorespiratory glycolate pathway (5), while one third of 14C-assimilates can be eliminated as a part of 14СО2 (6). A final content of bound 14C also depends on the amount of 14СО2 remaining in the leaf during the time between removing the photosynthetic chamber and fixing the leaf in alcohol, while the latter depends on degree of stomatal opening.
14C-carbon included in the final photosynthesis product – sucrose – in following 24 hours was exported from a donor leaf and distributed in different organs. The nature of this distribution was affected by light exposure as well (Fig. 2). The greatest export of 14С-assimilates from donor leaves occurred in medium light and minimum – in high light. The export of photosynthesis products from leaves was well correlated with rate of a downflow of assimilates. The ratio of radioactivity in plant parts above and below a donor leaf was greatest in medium light and minimum – in high light (difference between variants - more than 3-fold); plants grown in a faint light showed an intermediate value. At the same time, the content of labeled photosynthesis products in shoot apexes and tubers reliably differed in plants grown in low and high light (against the generally intense upward transfer of assimilates). In the first variant, the content of 14С in shoot apexes was almost the same as in a medium light, and 2-3 times higher under intense light. The content of 14С products in tubers was almost twice lower in high light than in other two variants (Fig. 2). There are several reasons for such different distribution of 14С in organs of plants grown in different light conditions. Firstly, intense light provides sufficient energy supply for photosynthesis and transfer processes, so resulting photosynthesis products enter the phloem to be delivered from leaves to consuming organs. At the same time, excessive illumination and overheating of plants contribute to increased transpiration.
Circulation of assimilates in plants has been shown previously (7). From leaves they move down along the phloem of a stem. A part of sucrose from the phloem enters the apoplast and then is brought away by the upflow of transpirational water (8). When sucrose reaches upper leaves it re-enters the phloem and is re-exported with a downflow. In the apoplast sucrose is partially hydrolyzed by invertase to products (glucose and fructose) utilized by cells. These processes are most intense in juvenile leaves not exporting assimilates due to underdevelopment of the phloem conduction system, so these leaves will accumulate 14С-labeled carbon. In this experiment, in a high light the increased upflow of transpirational water stimulated the upward transfer of 14C-sucrose received by the apoplast from stem phloem. This fact was suggested by the increased content of 14С-carbon in juvenile leaves of shoot apexes utilizing sucrose as well as in the donor leaf leaflets not exposed to 14СО2. In a photosynthetic chamber, 14С was absorbed by an only leaflet of a complex leaf; along the phloem of its petiole, 14С-sucrose was transferred to the apoplast and then returned in a backflow of transpirational water. As a result, a part of the labeled assimilates re-entered the leaflet – donor of 14C, as well as in other leaflets of this complex leaf.
The entry of labeled photosynthesis products to other (non-assimilated 14СО2) parts of 14С-donor leaf was almost similar in both variants of high and low light. In medium illumination, accumulation of the label was significantly lower. The environment under two layers of gauze contributed to a minimal transpiration and the transfer of 14С-labeled photosynthesis products in an upflow of water. This assumption was confirmed by a low content of 14С in shoot apexes of these plants. However, the content of label in neighboring leaflets of a donor leaf did not differ from that detected in conditions of intense light. Therefore, in plants exposed to a faint light, 14С-label was brought from a donor leaflet into adjacent cells through a symplastic pathway.
In low light, insufficient energy supply reduces the phloem transfer of assimilates, which contributed to a gradual diffusion of labeled assimilates from a donor leaflet through the symplast to neighboring parts of the leaf.
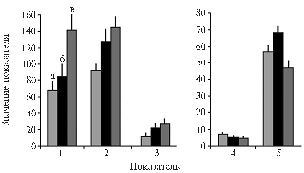 |
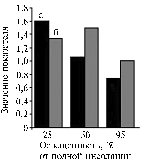
Fig. 3. Productivity characteristics of potato cv Nevsky grown in different light conditions; a, б , в — respectively, 25, 50 and 95 % of total insolation: 1 and 2 — resp., weight of potato tops and tubers, g/plant; 3 – average weight of one tuber, g, 4 - number of tubers, pcs., 5 – T, % (weight of tubers relative to a total plant biomass). Denotations: |
The transfer of labeled photosynthesis products to a shoot apex is complicated by a long distance. Besides, insufficient light and resulting energy deficit contribute to inhibition of nitrate reduction in roots, so more nitrates enter the leaves. This reduces conductivity of the phloem and, as a result, the outflow of assimilates from leaves (9). So, a high content of 14С-assimilates was delayed in leaves (both above and below a donor leaf). During an increased nitrate supply, hydrolysis of sucrose occurs much more intense in the apoplast than in mesophyll cells (10). Resulting hexoses can’t enter the phloem and have to return in mesophyll cells to be utilized there. In conditions of low transpiration, the absence of active circulation of assimilates within a plant eventually leads to less intense utilization in aboveground organs while a relatively more successful (but slower) symplastic transport of photosynthesis products to tubers. This process is quite energy-cost under low conductivity of the phloem, which was reflected by final productivity indices.
|
Fig. 4. Efficiency of utilizing PAR, g/W (а a) and photosynthesis in formation of tubers, g/g tops (б b) in potato cv Nevsky grown in different light conditions Denotations: |
Although the almost similar initial absorption of 14СО2 in leaves of different variants, light deficit has led to the minimum resulting productivity. Total weight of plants (Fig. 3) was generally correlated with amount of illumination. In the variant of excessive light, the weight of aboveground parts significantly exceeded that in other two variants which developed almost close values. At the same time, the weight of tubers in plants grown in medium and high light was almost similar. Ultimately, the efficiency of photosynthesis (weight of tubers formed per unit mass of photosynthesizing aboveground part of a plant) was found to be maximum in medium light and minimum – in high illumination. Plants grown in low light showed the intermediate efficiency of photosynthesis. This regularity was reflected by the value of Te % (percentage of economically important part of a harvest – tubers – relative to a total plant biomass) as well (Fig. 3). Decrease in Te was accompanied by reduce in efficiency of photosynthetic utilization of PAR (dry matter weight formed per 1 W absorbed light energy) (Fig. 4), although a high yield of tubers was obtained owing to extensification of the production process. Economically important part of a harvest is formed by three mechanisms of assimilates transfer combined in an optimum regime. In this experiment, the best weight of tubers per unit of absorbed light energy was obtained in medium illumination (Fig. 4).
Thus, growing potato in different light conditions has shown the influence of light intensity on the upflow transfer of assimilates with transpirational water and circulation within a plant. The enhanced upflow transfer contributes to intense utilization of photosynthesis products spent on formation of aboveground organs, which reduces Te and relative efficiency of photosynthesis, though the yield of tubers remains relatively high.
REFERENCES
1. Mokronosov A.T., Fotosinteticheskaya funktsiya i tselostnost’ rastitel’nogo organizma (Photosynthetic Function and Integrity of Plant Organism), Moscow, 1983.
2. Chikov V.I., The Role of Donor-Acceptor Relationship in the Regulation of Production Processes of Plants, in Fotosintez: fiziologiya, ontogenez, ekologiya (Compilation of Sci. Works: Photosynthesis: Physiology, Ontogenesis, Ecology), Ron’zhina E.S., Ed., Kaliningrad, 2009, pp. 206-234.
3. Mokronosov A.T., Photosynthesis in Potato, in Fiziologiya sel’skokhozyaistvennykh rastenii (Physiology of Agricultural Plants), Moscow, 1971, vol. 12, pp. 99-128.
4. Chikov V.I., Chemikosova S.A., Nesterova T.N. and Zernova O.V., Photosynthesis and Export Function of Leaves in Plants under the Increased Nitrogen Supply, in Fotozintez i produktsionnyi protsess (Compilation of Sci. Works: Photosynthesis and Production Process), Mokronosov A.T., Ed., Sverdlovsk, 1988, pp. 145-154.
5. Chikov V.I., Photorespiration, Sorosovsky obrazovatel’nyi zhurnal, 1996, vol. 11, pp. 2-8.
6. Laysk A.Kh., Kinetika fotosinteza i fotodykhaniya С3-rasteniy (Kinetics of Photosynthesis and Photorespiration in C3-Plants), Moscow, 1977.
7. Chikov V.I., Evolutionof Notions about Relationships between Photosynthesis and Plant Productivity, Fiziologiya rastenii, 2008, vol. 55, no. 1, pp. 140-154.
8. Minchin P.E.H. and McNaughton G.S., Xylem Transport of Recently Fixed Carbon within Lupine, Aust. J. Plant Physiol., 1987, vol. 14, pp. 325-329.
9. Batasheva S.N., Abdrakhimov G.G., Bakirova G.G. and Chikov V.I., Effect of Nitrates Supplied with the Transpiration Flow on Assimilate Translocation, Fiziologiya rastenii, 2007, vol. 56, no. 3, pp. 421-431.
10. Chikov V.I., Avvakumova N.Y., Bakirova G.G. et al., Apoplastic Transport of 14C-Photosynthates Measured under Drought and Nitrogen Supply, Biol. Plantarum, 2001, vol. 44, pp. 517-521.
1Kazan Institute of Biochemistry and Biophysics, RAS, Kazan 420111, The Republic of Tatarstan, Russia, |
Received May 5, 2010
|